
The ideal gas law treats gas molecules as point particles that interact with their containers but not each other, meaning they neither take up space nor change kinetic energy during collisions (i.e. In chemistry and thermodynamics, the Van der Waals equation (or Van der Waals equation of state) is an equation of state which extends the ideal gas law to include the effects of interaction between molecules of a gas, as well as accounting for the finite size of the molecules. ( Learn how and when to remove this template message) WikiProject Physics or WikiProject Chemistry may be able to help recruit an expert.  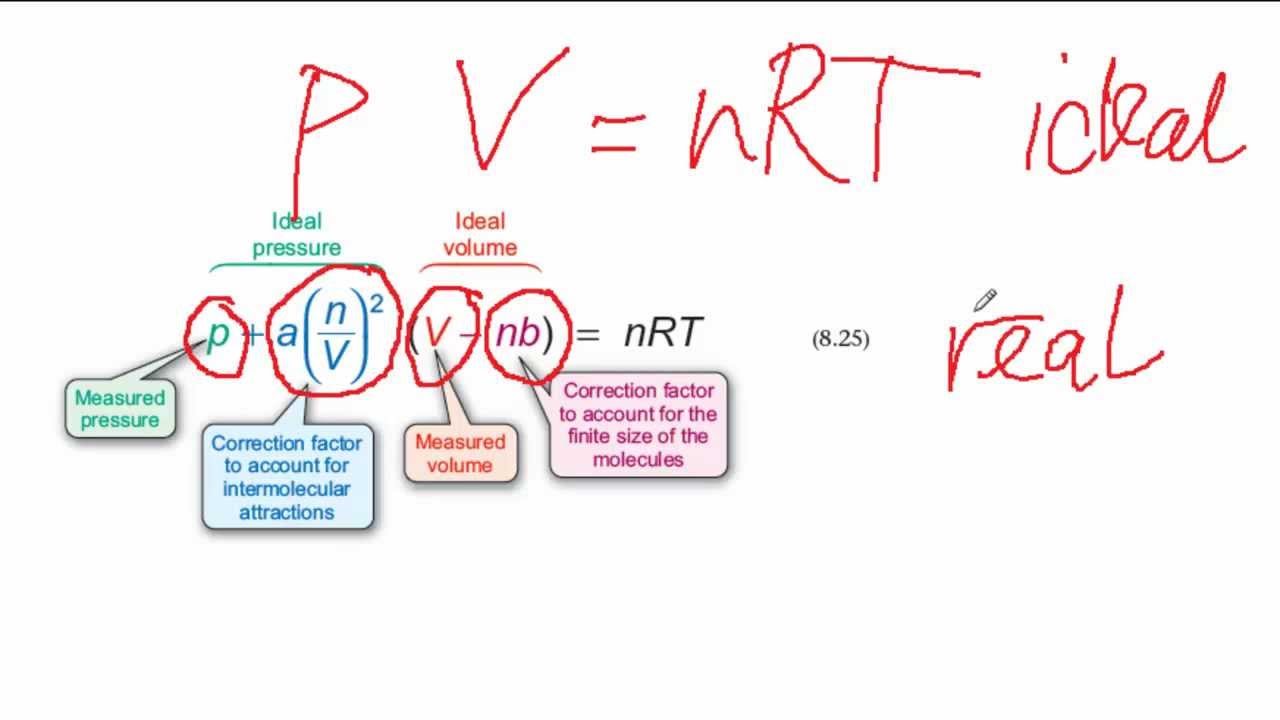
If a gas behaves ideally, both a and b are zero, and van der Waals equations approaches the ideal gas law PVnRT. They have positive values and are characteristic of the individual gas. Van der Waals Equation Calculator Water Heating Calculator. What is the percent difference between the pressure calculated using the van der Waals equation and the ideal pressure For N gas, a 1.390 L²atm/mol² and b 3. It is usually referred to as the van der Waals equation of state: P + a(n/V)2 (V/n - b) RT The constants a and b are called van der Waals constants. The specific problem is: the derivations are unsourced, and the text needs review by a physical chemistry expert to determine attribution of content. Ecuación de Van der Waals (RTemperatura/ (Volumen molar-Constante de gas b))- (Constante de gas a/Volumen molar2) p (RT/ (Vm-b))- (Ra/Vm2) Esta fórmula usa 1 Constantes, 5 Variables Constantes utilizadas R - Universal gas constant Valor tomado como 8. Find a Comprehensive Collection of Physics Calculators available for free and make your. Percent difference Pideal P van der Waals Pideal+Pvan der Waals 2 x 100. The ‘a’ and ‘b’ stands for constants specific to certain gases. What is the percent difference between the pressure calculated using the van der Waals equation and the ideal pressure For N gas, a 1.390 L²atm/mol² and b 3.910×10-² L/mol. Although it isn't trivial in general, you can check how the formula simplifies for processes mentioned below.This article needs attention from an expert in Physics or Chemistry. The Van der Waals equation can be written in this way: (P+an/V) (V-nb) nRT So in this equation P, V, R, T, and n stand for pressure, volume, gas constant, temperature, and moles of gas, respectively. The general formula for work done by the gas is expressed as ∫p(V)dV if we consider pressure as the function of volume. These parameters in real gases differ from theoretical ones, but we already contain them in our thermodynamic processes calculator. R for gases with more complex molecules.ΔT, where Cv is molar heat capacity under constant volume.Internal energy change is proportional to temperature variation ΔT and type of gas with the following equation: ΔU = Cv Still, it is possible to find thermal energy changes ΔU, which are described by the first law of thermodynamics: ΔU = Q - W, where Q denotes heat absorbed, and W is work done by gas. To use this online calculator for Van der Waals equation, enter Temperature (T), Molar Volume (Vm), Gas Constant b (b) & Gas Constant a (Ra) and hit the calculate button. Part of your confusion is in the formula you have written. It's quite tricky to estimate the precise value of internal energy. For a van der Waals gas, isothermal compressibility is V 2 ( V n b) 2 n R T V 3 2 a n 2 ( V n b) 2 If one substitutes the values of critical temperature and volume in this formula, isothermal compressibility goes to infinity. Internal energy U is the sum of all kinds of energy present in a system.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
